Ultimate Guide: Content Uniformity Test in Pharmaceutical Analysis USP<905>
What is Content Uniformity in Pharma?
Content uniformity is a standardized test that verifies every tablet, capsule, or unit dose delivers consistent amounts of the active pharmaceutical ingredient (API).
Content uniformity involves assaying each individual unit using an analytical method and comparing the results against established acceptance criteria from the USP.
How the Content Uniformity Test Works
Solid Dosage Forms
Assay 10 units individually using an appropriate analytical method
Calculate the acceptance value
Liquid Dosage Forms
Assay 10 units individually using an appropriate analytical method
Perform assay on the well-mixed material removed from an individual container in conditions of normal use
Report results as delivered dose
Calculate the acceptance value
Requirement to pass content uniformity
Acceptance value must be less than or equal to L1%
If acceptance value > L1%, test additonal 20 units and calculate acceptance value. The requirements are met if the final acceptance value of the 30 dosage units is ≤ L1%, and no individual content of any dosage unit is less than [1 − (0.01)(L2)]M nor more than [1 + (0.01)(L2)]M
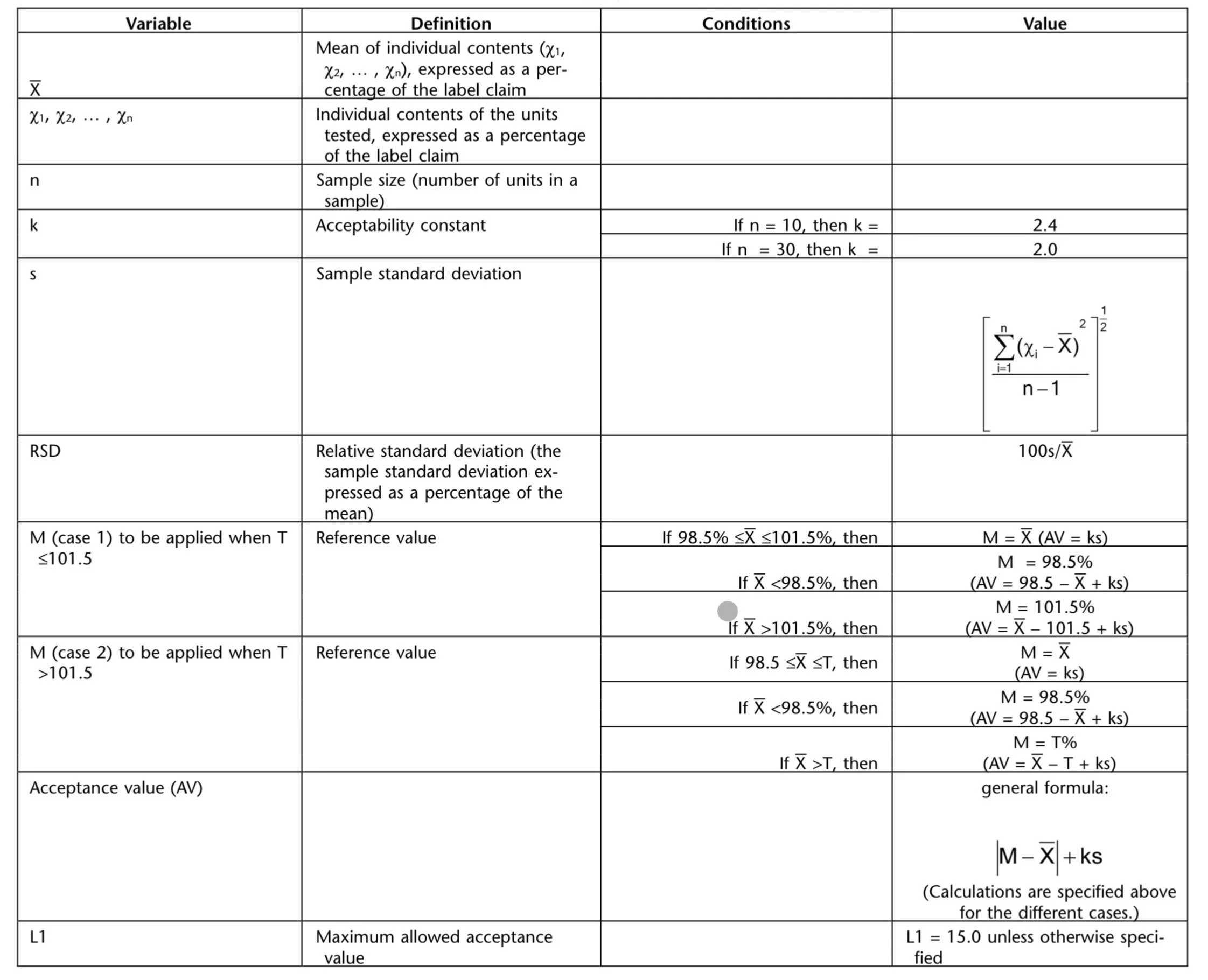
The acceptance value is |M-X| + ks, with the terms defined in the table below from USP <905>.
Reference: USP <905) Uniformity of Dosage Units
Read related article on Weight Variation in Pharmaceutical Analysis.
Feeling overwhelmed? Simplify the process with our content uniformity calculator. Quickly perform these calculations and make your life easier!
Content Uniformity Calculator Preview
Enter the % Label Claims for 10 dosage units, and the template will automatically calculate the acceptance value. It will also indicate whether the data passes or fails the test based on USP <905>.
Frequently Asked Questions
-
Content uniformity is a test described in USP <905> that evaluates whether individual dosage units contain the intended amount of API. The test ensures that each unit in a batch delivers a consistent dose to the patient.
-
10 dosage units are tested first. If the batch fails, 20 additional units are tested (total of 30).
-
The value of k depends on the number of dosage units tested. For a sample size of 10, k = 2.4, while for a sample of 30, k = 2.0
-
The reference value (M) represents the target content used in the Acceptance Value calculation. It is typically determined based on the mean of the sample results relative to the labeled claim.
In many cases:
If the mean result is between 98.5% and 101.5%, then M equals the sample mean.
If the mean falls outside this range, M is set to the nearest limit.
-
USP <905> also sets limits on individual dosage units:
No individual unit should be outside 75–125% of label claim.
In Stage 2 testing, not more than one unit may fall outside 85–115% of label claim.
These criteria ensure that extreme variability does not occur even if the AV passes.
-
Both tests evaluate dosage unit consistency but differ in approach:
Content Uniformity
Measures actual drug content in each unit
Used for low-dose formulations
Weight Variation
Measures total unit weight
Used when the API represents a large proportion of the dosage form